WHAT IS SMA® ALTHÉRA®?
Althéra®is an extensively hydrolysed (eHF) hypoallergenic infant formula that provides first-line relief from the symptoms associated with mild-moderate cows’ milk protein allergy (CMA) and/or multiple food protein allergies. SMA® Althéra® is nutritionally complete* and contains lactose to support growth and development. With its very low allergenic profile and preferred taste over casein-based eHFs, it is designed to support increased acceptability and tolerance in the majority of babies with CMA. Available in a 400g resealable tin. Free from soy.
WHO CAN USE SMA ALTHÉRA?
Babies and infants with cows’ milk protein allergy (CMA) can use Althéra formula.
For more information on SMA® Althéra® or cows milk allergy:
- Visit our CMA information page
- Contact our Careline on 0800 0 81 81 80
We have updated the preparation instructions on SMA® Althéra® to make them clearer to parents and caregivers. Both SMA® Althéra® and SMA® Alfamino® now have the same preparation instructions:
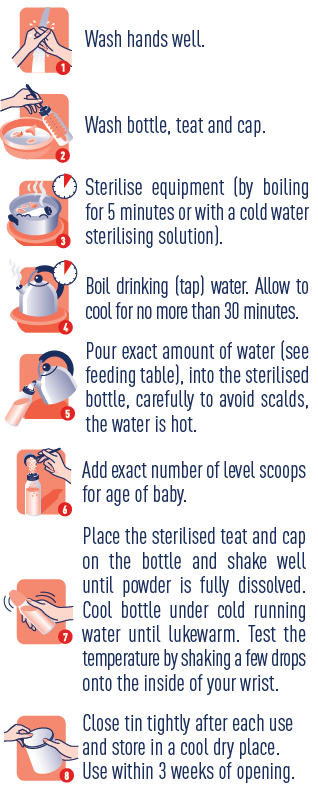
SMA® Althéra® is for complete nutritional support from birth or supplementary feeding from 6 months and up to 3 years of age for the dietary management of cows‘ milk allergy and/or multiple food protein allergies.
Precautions
Use under strict medical supervision after full considerations of all the feeding options available, including breastfeeding. For oral or enteral use only.
Lactose, maltodextrin, vegetable oils (sunflower, rapeseed, coconut), hydrolysed whey protein (milk), minerals (calcium glycerophosphate, potassium phosphate, magnesium chloride, calcium chloride, sodium chloride, potassium chloride, ferrous sulphate, sodium phosphate, zinc sulphate, potassium citrate, copper sulphate, manganese sulphate, potassium iodide, sodium selenate), emulsifier (E 472 c), Crypthecodinium Cohii oil (DHA), Mortierella Alpina oil (ARA), choline bitartrate, acidity regulator (E 330), vitamins (C, E, niacin, pantothenic acid, riboflavin, A, thiamin, B6, folic acid, K, D, biotin, B12), L-arginine, L-histidine, taurine, inositol, L-carnitine
Important notice:
Breastfeeding is best for babies. SMA® Althéra® must only be used under strict medical supervision and after full consideration of the feeding options available, including breastfeeding. The advice of a doctor, midwife, health visitor, public health nurse, dietitian or pharmacist on the need for and proper method of use of infant milks and on all matters of infant feeding should be sought. Good maternal nutrition is important for the preparation and maintenance of breastfeeding. Introducing partial bottle-feeding may have a negative effect on breastfeeding and reversing a decision not to breastfeed is difficult. Social and financial implications should be considered when selecting a method of infant feeding. Infant milk should always be prepared and used as directed. Inappropriate foods or feeding methods, or improper use of infant formula, may present a health hazard.
RELATED PRODUCTS
Learn more
Next, check out Compleat Paediatric® – a blenderised, nutritiously complete product which is great for tube feeding.
FAQs
Q: Does SMA® Althéra® contain lactose?
A: Yes, SMA® Althéra® is nutritiously complete and contains lactose.
Q: Is SMA Althéra® an alternative for breastfeeding?
A: Breastfeeding is best for babies and full consideration of all other feeding options must be considered. SMA Althéra® must be used under strict medical supervision, so we suggest consulting a professional.

We believe breast milk is the best food for infants. When in consultation with their healthcare professional, mothers and families find that optimal breastfeeding is not possible due to their infant’s medical condition, formulas for special medical purposes play a vital role in providing essential nutrients to infants. We have a global commitment to market breast-milk substitutes responsibly.
This website is about the management of cows’ milk protein allergy and nutritional solutions intended for infants. By continuing on this website, you accept that Nestlé Health Science supplies the information at your own request.

